Sustainability in medical device design and production
Throughout history, medical devices have always been considered as fundamental for the existing and future of human being, as they can help build healthier populations, solve health emergencies and advance the universal health system. Towards a more sustainable future for all, pursuing sustainability in the design and production of medical devices is equally important, hence challenging suppliers to be more innovative and responsible throughout their whole operation process.

The environmental problem of medical device
According to WHO, a medical device is defined as “any instrument, apparatus, implement, machine, appliance, implant, reagent for in vitro use, software, material or other similar or related article, intended by the manufacturer to be used, alone or in combination for a medical purpose”. In the UK, 66% of the NHS’s total carbon emissions is due to its supply chain, including pharmaceuticals, medical and non-medical devices.
Due to the special and strict requirements of the industry, it is generally simpler and more cost effective to discard most medical devices, as it is more complicated and expensive to go through the whole process of cleaning, sterilizing and disinfecting. In addition, while medical device producers have been attempting to meet the market’s rising demand for environmental sustainability, they must also address a critical component of their mission in ensuring the safety and usefulness of healthcare personnel and patients, so many devices frequently incorporate single-use throwaway plastic parts. Hence, a high amount of medical device waste is made up of single-use components and devices.
All stages of the medical device manufacturing, usage and disposal can have long-term environmental consequences. The carbon footprints are increasing as a consequence of the procurement of raw materials, the manufacture and transportation of medical equipment, the decontamination, repair, and sterilisation of medical devices and the disposal of medical devices and packaging, particularly from single-use plastics incineration. Medical device waste has also become a global issue as devices normally get more complicated and integrate more electronic components and diverse materials, making it much more difficult and expensive to be recycled and treated.
In 2017, out of 5.3 million tonnes of hazardous waste collected in England, 35% was recovered, 21% was transferred, 23% was treated, 6% was incinerated and 14% was sent to landfill, especially, the amount sent to landfills were significantly lower compared to the data in 2000, proving potential opportunities to tackle the problem of medical device waste.
Sustainability in medical device
While many people identify sustainability solely with environmental protection, sustainability actually encompasses far more than simply environmental concerns, as its pillars are based on environmental, social and economic sustainability. According to the Brundtland Commission of the United Nations in 1987, sustainability is defined as “meeting the needs of the present without compromising the ability of future generations to meet their own needs”. By pursuing sustainability in medical device design and production, manufacturers could help their sustainable medical devices become more environmentally friendly and appealing to customers, hence they can achieve cost savings, investor appeal, and increased brand and competitive advantage.
The sustainability of a medical device does not only involve the end of its lifecycle, whether it is recyclable or reusable, but instead, starts at the very beginning – the design phase. As around 80% of all product-related environmental consequences are determined during the product’s design phase, the early decisions on the design of the product have the most influence on the sustainability throughout its lifecycle. Moving on, the sustainability aspect also includes other factors like the potential to decrease carbon emissions, energy consumption, water use and material waste across the device’s full lifespan, from design and material selection to supply chain management, manufacturing and distribution.
For example, when it comes to deciding on the material for medical devices, designers and engineers need to consider criteria including renewability, resource efficiency, emission, raw material resources (ethical and environmental aspects), transportation, degradability and reusability. In recent year, some popular alternatives to traditional plastics are biopolymers polyhydroxyalkanoates (PHA), which do not induce allergic responses and are biodegradable in the body, and polylactide (PLA), a material generated from maize starch that is similarly more readily biodegradable than crude oil-based plastics. By working with tool manufacturers and suppliers, clients can specify their choice of materials to renovate the sustainability aspect of their products.

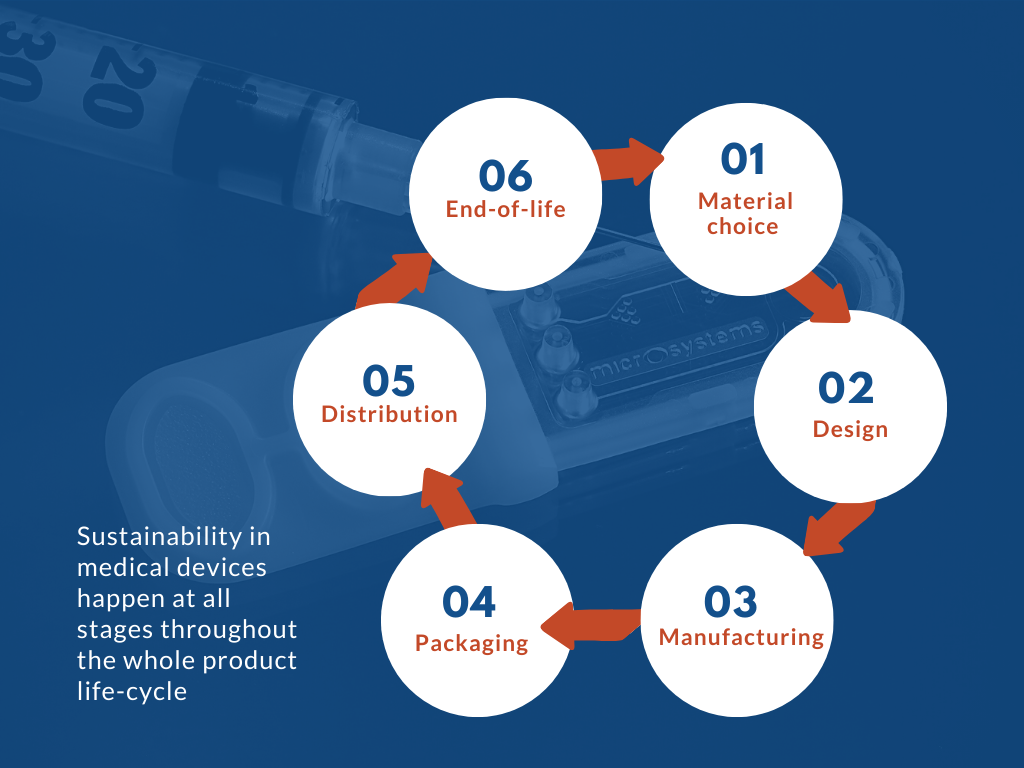
In general, medical device designers, manufacturers and users should constantly challenge themselves throughout all stages of the products’ life cycles:
- Material choice : Medical device’s material choice could significantly decide the overall impacts throughout its lifecycle, as it directly impacts all the other stages. For example, while plastic is advantageous in its lightweight (hence lighter for transportation), high durability (long lifecycle with less maintenance), it can take between 20 to 500 years to degrade, depending on the nature of the material and environmental conditions such as sunshine exposure. Material choice should be carefully decided, based on its fitness for purpose, environmental impact, recycled content, local context, responsible sourcing and end-of-life treatment.
- Can a petroleum-based plastic be replaced with a bio-based material?
- Should the number of different materials utilised be reduced to make the device easier to be recycled at the end of its lifecycle?
- Should the plastic part be replaced with glass or stainless steel so that it can be repurposed and reused?
- Where does the material come from? How transparent is the material? Is the material available locally? Who are involved in the production of the material?
- Design : Most medical devices are becoming smaller and smarter, to meet the more advanced application requirements from the market, at the same time, to reduce cost, raw material and processed resources. By practising design for sustainability, businesses could reduce negative environmental impacts, energy and natural resource usage, and implement socially responsible practices for employees, local communities and the society across the whole product and service life cycle.
- Can I decrease the overall size or weight of the device to reduce material consumption, packaging, and carbon emissions associated with distribution?
- Can the design be improved to increase the lifespan of the device? How durable is the design?
- How to make the device easy to maintain?
- Can electronic components be shrunk to lessen their final disposal impact?
- Is it possible to lower the battery size or convert to a more sustainable battery technology (for example, rechargeable batteries)?
- How to reduce the amount of different materials used?
- Manufacturing : It is crucial for manufacturers to measure, analyse and improve their technologies and processes, such that they can control their impacts on the environment, while increasing productivity and reducing time to market, overall helping them to achieve the device’s long-term environmental effect.
- Is it possible to manufacture in a way that saves energy and water?
- Is it possible to employ additive manufacturing to decrease material waste?
- Is my contract manufacturer adhering to environmental best practices for carbon capture and waste stream disposal?
- Can I improve the manufacturing process with my current resources?
- Are my employees satisfied with their well-beings, safety and working environment throughout the operation process?
- Packaging : Packaging has significant environmental impacts, which encourages most businesses to reduce their use of it.
- How can I reduce packaging waste?
- Is it possible to utilise recyclable or biodegradable packaging materials?
- Is it possible to remove the usage of plastic or petroleum-based foam packing elements?
- Can my customers cooperate with me to reduce or return or reuse the packaging waste?
- Can I move from paper-based documentation to on-device content when practical?
- Distribution : Distribution is critical when attempting to reduce the overall carbon impact, however, it is extremely challenging in a global market where products and raw materials are generally imported. Businesses could consider prime production and storage locations that minimise long-distance traffic, and the use of new technology like electric cars.
- Can production and warehouse sites be selected to reduce long-distance transportation?
- How can I design the distribution schedule to maximise transportation efficiency?
- How can I work with my customers to improve the current distribution?
- End-of-life : The requirement to meet high safety standards limits efforts to minimize the amount of disposable components. The concerns involved with hazardous medical waste and biological contamination, as well as the expensive expense of product sterilization and reprocessing, have hindered many businesses and customers from transitioning from disposable to more sustainable devices.
- Is it possible to make the transition from a single-use device to a reusable device?
- Can the device or its components be recycled? Could it be returned to the manufacturer for reconditioning?
- If thrown away, what are the environmental impacts of the device? How can I minimize that?
- Do I need special waste treatment service for the device?
Besides, medical device businesses also need to look into other aspects related to its structures, including overall environmental footprint (office waste, sourcing energy sustainably, etc.), social responsibility (health and safety, training, diversity, equality, community giveback, etc.).
Several issues must be considered while developing medical devices, including safety, usage, dependability and durability. Medical device sustainability is increasingly being added to the list of considerations that manufacturers must address, to not only meet the new demands from global markets, but also to achieve a sustainable growth for the businesses themselves. The dedicated teams of experts at Micro Systems are committed to supporting our customers and business partners in integrating sustainability throughout the whole lifecycle of our medical devices. We work alongside with our customers from the very first step of the designs, with the desire to innovate and integrate the latest technologies in our solutions, to achieve breakthroughs with sustainability in the world of medical devices.
Check out our 2022 CSR Report here!
Contact us today at info@microsystems.uk.com to discuss with our experts at Micro Systems (UK).

Micro Systems specializes in the design, manufacture and validation of ultra precision micro molds for the medical, pharmaceutical and optical markets, at the same time, the development and use of micro and nano technologies in the design and manufacture of injection molded components. We have a dedicated micro molding facility, and have ISO13485 and ISO9001 certifications. For more information, please Contact us or visit our website.